

← Home • Products • Reverse Osmosis ↓ Summary • Design Example • Process Description • Applications • Technical Data • Downloads
Reverse osmosis (short: RO) is a membrane process, which in water treatment is used for →demineralisation, meaning for removal of dissolved solids from water.
For demineralisation, water at high pressure flows across the surface of a semi-permeable membrane arranged within membrane elements. The semi-permeable membrane separates the membrane elements into a concentrate side and a permeate side. The pressure differential between the concentrate side and the permeate side results in water flowing from the concentrate side and through the semi-permeable membrane into the permeate side, while the most dissolved solids remain on the concentrate side.
Reverse osmosis is a continuous process. During service, concentrate will continuously be discharged as waste water.
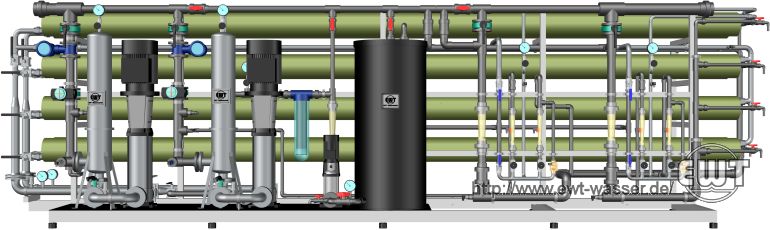
Reverse osmosis plant, two lines, single pass, concentrate-staged, with integrated cleaning station.
Osmosis occurs if two aqeuous solutions of different mineral concentrations are separated by a semi-permeable membrane. This system aims to balance the mineral concentrations between the two solutions. A semi-permeable membrane allows the passage of water, but blocks the passage of minerals. Accordingly, water molecules flow from the more dilute solution and through the semi-permeable membrane into the less dilute soltion. The resulting increase of the amount of water molecules in the less dilute soltion in turn results in pressure acting from the side of the less dilute solution onto the side of the more dilute solution. This pressure is called osmotic pressure. The osmotic pressure hinders the flow of water molecules through the semi-permeable membrane. The system aims towards a state of equilibrium; further dilution of the less dilute solution results in an increase of osmostic pressure, while at the same time the concentration difference is reduced. The osmotic pressure and the concentration difference will eventually reach a state of equilibrium, the system will then not change any further without outside influence.
By enacting a pressure in direction of the concentration gradient, meaning onto the more concentrated solution, the state of equilibrium as described above will change. If the thus enacted pressure exceeds the osmotic pressure of the system at the state of equilibrium, the flow direction of the water will be reversed. Water will then flow from the more concentrated solution and through the semi-permeable membrane into the less concentrated solution. The mineral concentration of the more concentrated solution will then increase even further, while the less concentrated solution will be diluted further. This process is called reverse osmosis. The concentrated solution is called concentrate, while the diluted or demineralised solution is called permeate.
In a reverse osmosis plant, the working pressure acting reverse to the concentration gradient is generated by a high pressure pump. The feed flows through the high pressure pump and into the reverse osmosis modules. These reverse osmosis modules consist of a pressure vessel containing membrane elements, and three pipeline connections: one each for feed, permeate, and concentrate. The membrane elements consist of a core on the permeate side, and of a semi-permeable membrane arranged around this core on the concentrate side
The feed flows into the reverse osmosis module on the concentrate side. Accordingly, the working pressure generated by the high pressure pump acts from the concentrate side onto the permeate side. This results in a flow of water through the semi-permeable membrane and into the permeate side, according to the principle of reverse osmosis as described above. The majority of minerals diluted in the feed however remain on the concentrate side. Accordingly, the water on the permeate side is demineralised, while the mineral concentration of the concentrate side increases. Permeate and concentrate flow out of the reverse osmosis module separately, through their respective outlet connection.
Reverse osmosis is a continuous process, meaning that the feed will continuously flow into the reverse osmosis modules, and both permeate and concentrate will continuously flow out of the reverse osmosis modules.
The ratio of permeate volume flow to feed volume flow is defined as recovery. The differential between feed volume flow and permeate volume flow is equal to the concentrate volume flow. The differential between the total dissolved solids (TDS) of the feed and the TDS the permeate, divided by the TDS of the feed, is defined as salt rejection. The ratio of the TDS of the concentrate to the TDS of the feed is defined as concentrate cycles of concentration. In cases of high salt rejection, the concentrate cycles of concentration are approximately equal to the reciprocal value of the recovery.
As a result of the concentrate cycles of concentration, the concentration of certain dissolved solids in the concentrate may exceed the limit of solubility. This will lead to deposition of minerals on the membrane elements, called scaling. There may also be deposits on the membrane elements reulsting from suspended solids, organic compounds, or biological growth, called fouling. Scaling and fouling will reduce the effectiveness of membrane elements, and may permanently damage the membrane elements. In order to prevent or at least reduce scaling and fouling, the reverse osmosis feed is usually treated, for example by ion exchange →dealkalisation, →softening, or →chemical conditioning with antiscalant. Further, membrane aging will also gradually reduce the effectiveness of the membrane elements.
| permeate volume flow | available from approx. 0.5 up to 100 m³/h for each line | ||
| amount of lines | usually 1× 100% or 2× 50% | ||
| salt rejection | ≥ 96% ... 99.6%, single pass design | ||
| permeate quality | electrical conductivity |
≤ 30 μS/cm, brackish water or fresh water demineralisation ≤ 1000 μS/cm, sea water desalination |
|
| recovery |
usually 70% ... 90%, brackish water or fresh water demineralisation usually 30% ... 45%, sea water desalination |
||
| operating pressure |
approx. 5 ... 25 bar, brackish water or fresh water demineralisation approx. 60 ... 80 bar, sea water desalination |
||
| operating temperature | usually 5 ... 35 °C | ||
| waste water | corresponding to the recovery, for example 250 ... 333 L for each 1 m³ permeate with a recovery of 75% ... 80% | ||
| electrical energy demand for pressure boosting |
approx. 0.4 .... 1.5 kWh for each 1m³ permeate, brackish water or fresh water demineralisation approx. 3 .... 6 kWh for each 1m³ permeate, sea water desalination |
||
| recommended feed quality | turbidity | < 1 NTU | |
| total suspended solids (TSS) | < 0.5 ... 1 mg/L | ||
| Silt Density Index (SDI15 min) | < 3 ... 5 %/min | ||
| iron | < 0.05 mg/L Fe | ||
| manganese | < 0.05 mg/L Mn | ||
| aluminium | < 0.05 mg/L Al | ||
| free chlorine | < 0.1 mg/L Cl2 | ||
| hydrogen sulfide | < 0.1 mg/L H2S | ||
| total organic carbon (TOC) | < 3 mg/L C | ||
| oil, grease | < 0.1 mg/L | ||
| Further parameters (e.g. temperature, pH value, water hardness, chloride, sulphate, silicic acid) are each limited for a given recovery and feed quality. | |||
| membrane element cleaning intervals | usually approx. 6 ... 36 months | ||
| membrane element total service life | usually approx. 3 ... 5 years, in some cases up to 10 years | ||
| material options | membrane elements | • polyamide (PA) | |
| pressure vessels |
• glass-reinforced plastic (GRP) • stainless steel (e.g. 1.4404) |
||
| cleaning tank | • polyethylene (PE) | ||
| booster pump | • stainless steel (e.g. 1.4301, 1.4404) | ||
| pipelines |
• polyvinyl chloride (PVC) • polypropylene (PP) • polyvinylidene fluoride (PVDF) • stainless steel (e.g. 1.4404, 1.4539) |
||
| valves |
• polyvinyl chloride (PVC) • polypropylene (PP) • polyvinylidene fluoride (PVDF) • brass (e.g. CW617N, CW710R) • stainless steel (e.g. 1.4408) |
||
| gaskets |
• etyhlene propylene diene monomer rubber (EPDM) • polytetrafluorethylene (PTFE) • NBR composite |
||
| skid | • stainless steel (e.g. 1.4301) | ||
| control options | start-up, service, and shut-down |
• fully automated, via micro-processor control unit • fully automated, via PLC |
|
| cleaning of the membrane elements |
• manually • partly automated, via micro-processor control unit • partly automated, via control circuitry • partly automated, via PLC |
||
| process monitoring |
• gauge pressure (standard) • differential pressure (standard) • volume flow concentrate (standard) • volume flow permeate (option) • volume flow feed (option) • electrical conductivity permeate (standard) • pH value concentrate (option) • water hardness feed (option) • ... |
||
2018-05-05 • water treatment made in Germany • Company Information • Privacy