

← Home • Applications • Cooling Water ↓ Introduction • Make-up Water Treatment • Side Stream Filtration • Blow-down Control • Chemical Conditioning
For the reliable and economic operation of open circuit cooling systems, corrosion, scaling, and fouling need to be minimised while at the same time maintaining economic cooling water cycles of concentration. To this end, the make-up water and the cooling water are treated by various water treatment processes.
Simplified Flow Diagram of an Open Circuit Cooling System:
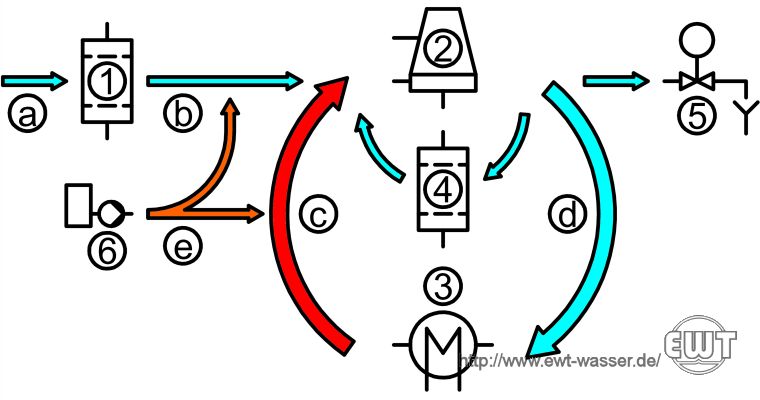
1 = make-up water treatment, 2 = cooling tower, 3 = process (e.g. heat exchanger), 4 = side stream filter, 5 = blow-down valve, 6 = dosing plant
a = raw water, b = make-up water, c = cooling water - hot, d = cooling water - cold, e = conditioning agents
The hot cooling water (c) flows from the process (3) through the cooling tower (2), and is cooled by evaporation. The cold cooling water (d) flows back through the process (2), and is heated up again. A part of the cooling water is circulated through a side stream filter (4), in order to limit the concentration of suspended solids. The cooling water cycles of concentration are adjusted by the blow-down unit (5). Water losses due to evaporation, drift and blow-down are replenished by make-up water (b), which is produced from raw water (a), for example surface water, in the make-up water treatment plant (1). For chemical conditioning, various conditioning agents (e) are added to the make-up water (b) and cooling water (c) by a dosing plant (6).
Corrosion, scaling, and fouling within the open circuit cooling system relate to the interdependency between dissolved solids, suspended solids, the cooling water temperature, biological growth, flow conditions, and the material of the different system components like pipelines, valves, heat exchangers, and cooling water basins. Together with the make-up water (b), both dissolved and suspended solids enter the cooling system, for example calcium, sodium, hydrocarbonate, sulphate, chloride, and also iron, manganese, or silicate compounds. In the cooling tower (2), suspended solids enter the cooling system with the air drift, furthermore, a balancing between the concentrations of gases in the air and gases dissolved within the cooling water occurs, for example oxygen and carbon dioxide.
While water evaporates in the cooling tower, both dissolved and suspended solids remain within the cooling system, leading to an increase of concentration. The concentration ratio between dissolved solids in the cooling water and dissolved solids in the make-up water is defined as cycles of concentration (CoC). Assuming a constant total cooling water volume, the cooling water cycles of concentration directly relate to evaporation, drift, and blow-down. For a given make-up water quality, given process conditions, and a given chemical conditioning program, there is usually a certain range of optimum cycles of concentration with minimal expected corrosion, scaling, and fouling. For economic reasons, system designers usually aim to achieve between 3 and 10 cycles of concentration.
The main goal of make-up water treatment is the removal of both dissolved and suspended solids from the raw water. Depending upon the raw water quality and source, the planned cooling water cycles of concentration, and any planned further use of the make-up water as process or production water, various filtration, ion exchange, or membrane separation processes can be used.
For example, either a →sand filter or a →multi-media filter is usually used for removal of suspended solids, as well as for removal of iron and manganese. For removal of calcium and magnesium ions, either a →dealkalisation plant or a →softening plant is used, while for demineralisation, a →reverse osmosis plant is used. In case of treatment by softening or reverse osmosis, it is commong practice to further blend the treated water with filtrated raw water, in order to achieve favourable chemical conditions regarding corrosion within the cooling system.

Side stream filter for cooling water, with integrated blow-down control.
Side stream filtration serves for limiting the concentration of suspended solids in the cooling water. Suspended solids enter the cooling system with the make-up water and cooling tower air draft, however they may also be a result of corrosion or biological growth. For most applications, limiting the concentration of suspended solids to < 10 mg/L is recommended; approximately 5 ... 10% of the cooling water volume flow are usually filtrated with a side stream filter for this purpose. In case of highly effective solid dispersants being used, or in case of automated cleaning mechanisms for important cooling system components, higher suspended solid concentrations may be acceptable.
For side stream filtration, a →sand filter is usually used. In case of less strict requirements regarding the suspended solids concentration, different kinds of self-cleaning surface filters are also used instead, for example disc filters or screen filters. In many cases, cooling water can be used for backwashing the side stream filter. The backwash effluent can in these cases be substracted from the cooling system blow-down, thus in many cases resulting in an effective backwash water consumption of zero, depending upon the backwash intervals of the side stream filter.
Cooling water blow-down serves for adjusting the cooling water cycles of concentration, as ↑defined above. Usually, blow-down occurs by automated opening and closing of the blow-down valve, triggered by set limit values for different chemical parameters of the cooling water, for example the →electrical conductivity. The blow-down valve can for example be located on the cooling water basin, or be together with the related instrumentation be integrated into the ↑side stream filter piping.
Depending upon the raw water quality, the expected cycles of concentration, and the type of chemical conditioning, it may in some cases be beneficial to automatically monitor further chemical parameters of the cooling water, in addition to the electric conductivity, for example the pH-value, the alkalinity, the iron concentration, or the concentration of certain conditioning agents.
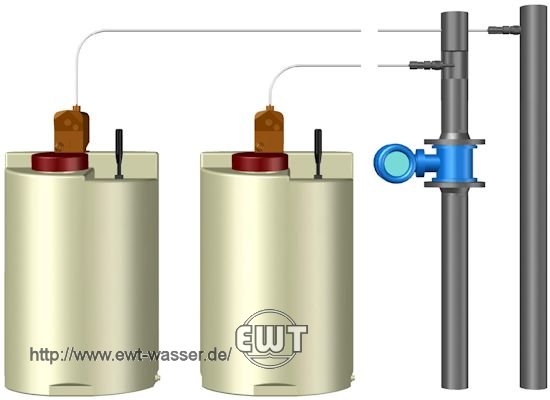
Dosing system for cooling water treatment with corrosion inhibitor and biocides.
The operation of open cooling systems usually requires the application of chemical conditioning programs. →Dosing systems are used for adding the chemical conditioning agents into the make-up water or into the cooling water at suitable injection points.
Commonly applied chemical conditioning programs include for example corrosion inhibitors, scale inhibitors, and biocides for limiting biological growth. In addition – or in certain cases as an alternative – acids, for example sulfuric acid, or caustics, for example sodium hydroxide, may be added to the make-up water or cooling water for pH-adjustment, in order to influence the carbonate chemistry.
2018-05-05 • water treatment made in Germany • Company Information • Privacy